
Figure 1
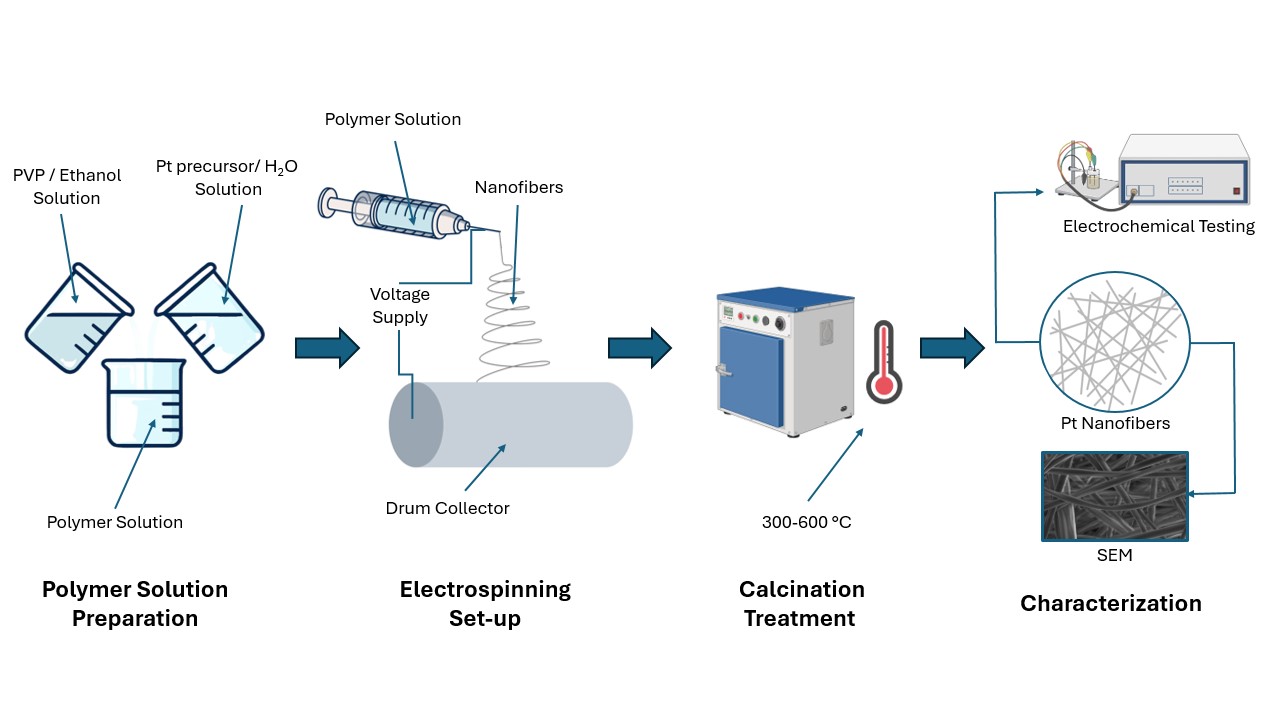
Figure 2

Team 05
Team Members |
Faculty Advisor |
Kinee Christian |
Dr. Jeffrey McCutcheon Sponsor Nel Hydrogen |
sponsored by
Sponsor Image Not Available
Synthesizing Catalyst Nanofibers for Hydrogen Electrolyzers
As fossil fuels are depleted and their harmful environmental impacts increase, there is a focus on utilizing renewable energy sources, such as hydrogen. Hydrogen plays a key role in the transition to cleaner fuels because it can store and deliver energy without producing greenhouse gas emissions. Technologies such as Proton Exchange Membrane (PEM) electrolyzers, can use renewable electricity to split water and produce hydrogen. Currently, platinum group metal catalysts are the industry standard for facilitating hydrogen production, but they can be extremely expensive and rare. Our team proposes using platinum nanofibers, an alternative catalyst shape (compared to typical nanoparticles), that reduce the required material loading while maintaining electrolyzer performance. This design increases the available surface area of the catalyst, allowing for higher reaction efficiency while using less material. Additionally, nanofibers offer improved mass transport and durability compared to nanoparticles. We chose to synthesize platinum nanofibers through electrospinning, as this fabrication method offers precise control over processing and fiber parameters, and thus their morphology. After electrospinning, we used calcination, a high temperature treatment, to transform initial fibers into stable and catalytically active materials. The performance of these catalysts will be evaluated in PEM electrolyzers and through a techno-economic analysis, to determine whether this design can outperform conventional platinum catalysts. By demonstrating superior electrolyzer performance, platinum nanofibers can address the economic constraints limiting electrolyzer commercialization and pave the way for more sustainable energy sources.
